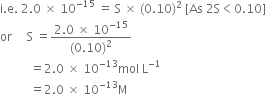SOLVED: The pH of a saturated solution of nickel hydroxide, Ni(OH)2, is 8.83. Calculate the Ksp for nickel hydroxide. (Show all relevant chemical reactions and work.)

Strongly Coupled Ni/Ni(OH)2 Hybrid Nanocomposites as Highly Active Bifunctional Electrocatalysts for Overall Water Splitting | ACS Sustainable Chemistry & Engineering

Pt and Pt–Ni(OH)2 Electrodes for the Hydrogen Evolution Reaction in Alkaline Electrolytes and Their Nanoscaled Electrocatalysts - Ruqia - 2018 - ChemSusChem - Wiley Online Library

Enhancing Hydrogen Evolution Activity in Water Splitting by Tailoring Li+-Ni (OH)2-Pt Interfaces | Science

Table 2 from Synthesis of Me Doped Mg(OH)2 Materials for Thermochemical Heat Storage | Semantic Scholar

Heterojunction α-Co(OH)2/α-Ni(OH)2 nanorods arrays on Ni foam with high utilization rate and excellent structure stability for high-performance supercapacitor | Scientific Reports

Heterojunction α-Co(OH)2/α-Ni(OH)2 nanorods arrays on Ni foam with high utilization rate and excellent structure stability for high-performance supercapacitor | Scientific Reports

Ionic product of Ni{(OH)}_{2} is 2.0times {10}^{-15}. Molar solubility of Ni {(OH)}_{2} in 0.10M NaOH will be ______.1.0times {10}^{-13}M4.0times {10}^{-13}M8.0times {10}^{-13}M2.0times {10}^{-13}M

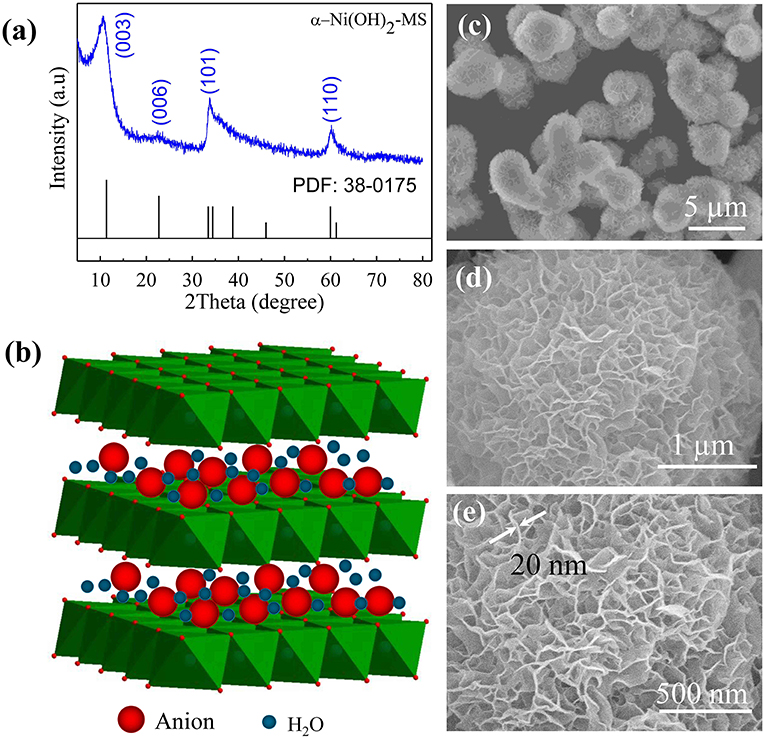
Figure 4 from Preparation and Characterization of Ni ( OH ) 2 and NiOMesoporous Nanosheets | Semantic Scholar

Find out the solubility of Ni(OH)_2 in 0.1 MNaOH. Given that the ionic product of Ni(OH)_2 is 2 ×... - YouTube
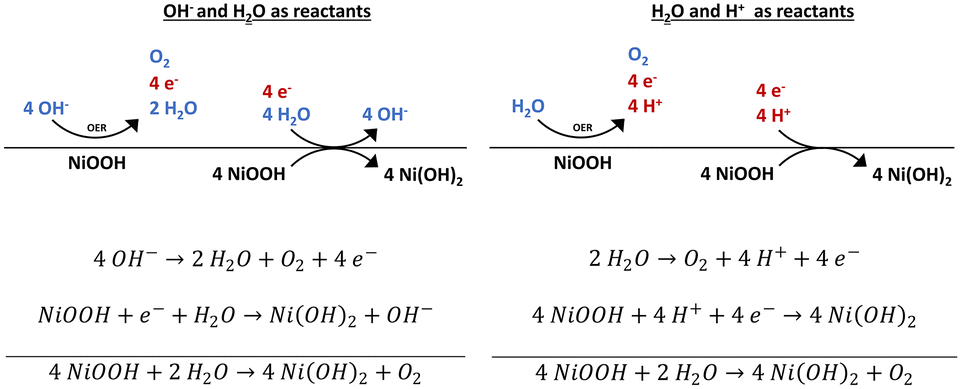
Stability and decomposition pathways of the NiOOH OER active phase of NiO x electrocatalysts at open circuit potential traced by ex situ and in situ s ... - Catalysis Science & Technology (

Interfacial electron rearrangement: Ni activated Ni(OH)2 for efficient hydrogen evolution - ScienceDirect

Interface engineering: The Ni(OH)2/MoS2 heterostructure for highly efficient alkaline hydrogen evolution - ScienceDirect

Bifunctional Tungsten-Doped Ni(OH)2/NiOOH Nanosheets for Overall Water Splitting in an Alkaline Medium | ACS Applied Nano Materials